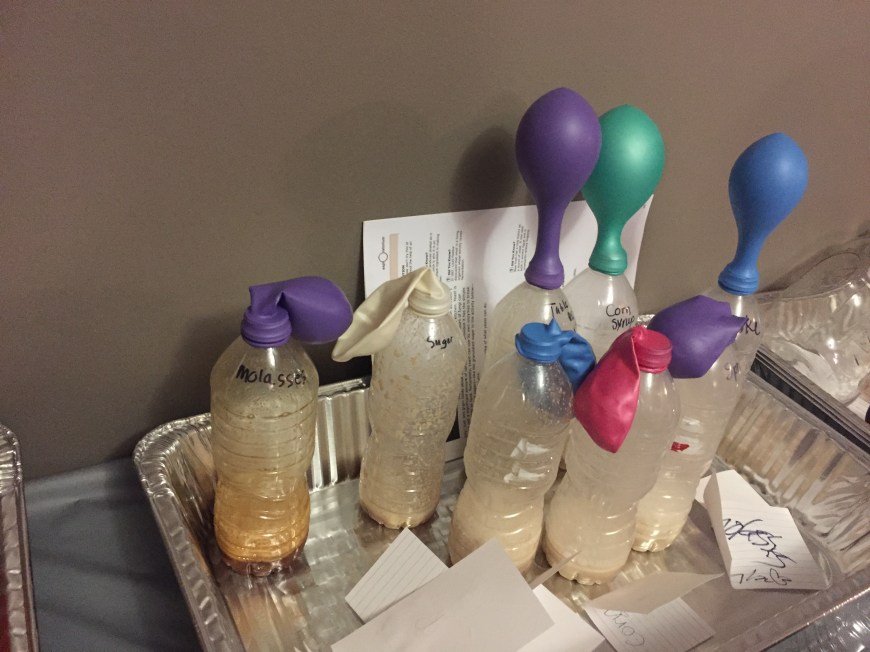
We did a couple of other experiments. We did the dancing gummy worms experiment. It was kind of a dud. We also did the Mentos and Diet Coke geyser. That was lots of fun!! Most of the adults had never seen it before, so it was really a treat for them. They also did the turn an egg shell into mercury glass. It was really tough and took forever to do. The ones that had the patience, did get to see how cool it turned out in the end. I also had prepared slides for them to look at under the microscope.


We played trivia of course. I had educational posters up all over the house. They were cute decorations, and now make their home in the school room. Back to trivia, most of the answers could be found on the posters.
We also made cells from cookies and candies. A few of them turned out really good!

I made chocolate lab rats, fossil cookies, petri dish jello, strawberries, pineapple, and grapes made up the fruit tray. We had a cheese tray. We had chips and guacamole and potato chips. We had tuna and chicken salad as well. I made educational signs for the foods and hung them by them.



I made a cake with sprinkles and a jello petri dish.

The invitation was from Etsy shop ohbejoyfulshop. 
It was such a fun party!